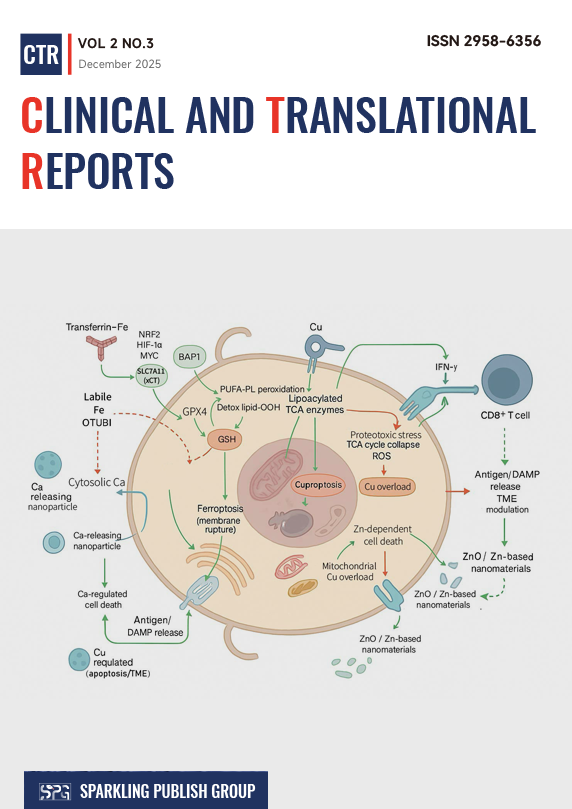
Metal ions such as iron, copper, calcium and zinc are indispensable for cellular metabolism, yet their local overload or mislocalization can trigger distinct forms of programmed cell death (PCD). Over the past decade, ferroptosis and cuproptosis have emerged as prototypical “metal ion-induced deaths”, while Ca²⁺- and Zn²⁺-associated death programs and multi-metal “metal overload” are beginning to be appreciated. This review summarizes the mechanisms and translational studies of metal ion-regulated cancer cell death, illustrating how these pathways are linked to tumor cell metabolism, plasticity, and the tumor microenvironment (TME). The dual nature of metal ion-induced deaths in cancer is explored here. As a tumor-suppressive mechanism engaged by tumor suppressors, immune effector cells and cytotoxic therapies, but also as a driver of chronic inflammation, clonal selection and therapy resistance when incompletely executed. Next, small molecules, metal-based nanomedicines and combination strategies that exploit ferroptosis, cuproptosis, Ca²⁺ overload or Zn-dependent death to overcome resistance and reshape the TME are evaluated in detail. Together, the evidence argues that “metal ion-induced deaths” constitutes a central node linking metabolism, stress signaling and immunity.
1. Mishra AP, Salehi B, Sharifi-Rad M, Pezzani R, Kobarfard F, Sharifi-Rad J, et al. Programmed cell death, from a cancer perspective: an overview. Molecular diagnosis & therapy. 2018;22(3):281-95.
2. Liu J, Hong M, Li Y, Chen D, Wu Y, Hu Y. Programmed cell death tunes tumor immunity. Frontiers in immunology. 2022;13:847345.
3. Wei S, Han C, Mo S, Huang H, Luo X. Advancements in programmed cell death research in antitumor therapy: a comprehensive overview. Apoptosis. 2025;30(1):401-21.
4. Hänggi K, Ruffell B. Cell death, therapeutics, and the immune response in cancer. Trends in cancer. 2023;9(5):381-96.
5. Garg AD, Agostinis P. Cell death and immunity in cancer: From danger signals to mimicry of pathogen defense responses. Immunological reviews. 2017;280(1):126-48.
6. Song Q, Yang Y, Yang L. Navigating Transition Metal‐Dependent Cell Death: Mechanisms, Crosstalk, and Future Directions. Advanced Science. 2025:e01974.
7. Parida L, Patel TN. Systemic impact of heavy metals and their role in cancer development: a review. Environmental Monitoring and Assessment. 2023;195(6):766.
8. Tang D, Kroemer G. Ferroptosis. Current Biology. 2020;30(21):R1292-R7.
9. Garza-Lombó C, Posadas Y, Quintanar L, Gonsebatt ME, Franco R. Neurotoxicity linked to dysfunctional metal ion homeostasis and xenobiotic metal exposure: redox signaling and oxidative stress. Antioxidants & redox signaling. 2018;28(18):1669-703.
10. Orellana-Urzua S, Rojas I, Libano L, Rodrigo R. Pathophysiology of ischemic stroke: role of oxidative stress. Current pharmaceutical design. 2020;26(34):4246-60.
11. Li Y, Du Y, Zhou Y, Chen Q, Luo Z, Ren Y, et al. Iron and copper: critical executioners of ferroptosis, cuproptosis and other forms of cell death. Cell Communication and Signaling. 2023;21(1):327.
12. Venkataramani V. Iron homeostasis and metabolism: two sides of a coin. Ferroptosis: Mechanism and Diseases. 2021:25-40.
13. Ajayi ND, Ajayi SA, Oladoyinbo OB, Olaniyi OO. A review of literature on transferrin: Deciphering its complex mechanism in cellular iron regulation and clinical implications. Available at SSRN 4690424. 2024.
14. Zhou N, Wei S, Sun T, Xie S, Liu J, Li W, et al. Recent progress in the role of endogenous metal ions in doxorubicin-induced cardiotoxicity. Frontiers in Pharmacology. 2023;14:1292088.
15. Yan H, Talty R, Aladelokun O, Bosenberg M, Johnson CH. Ferroptosis in colorectal cancer: a future target? British Journal of Cancer. 2023;128(8):1439-51.
16. Mortensen MS, Ruiz J, Watts JL. Polyunsaturated fatty acids drive lipid peroxidation during ferroptosis. Cells. 2023;12(5):804.
17. Ding K, Liu C, Li L, Yang M, Jiang N, Luo S, et al. Acyl-CoA synthase ACSL4: an essential target in ferroptosis and fatty acid metabolism. Chinese medical journal. 2023;136(21):2521-37.
18. Zhang X, Hou L, Guo Z, Wang G, Xu J, Zheng Z, et al. Lipid peroxidation in osteoarthritis: focusing on 4-hydroxynonenal, malondialdehyde, and ferroptosis. Cell death discovery. 2023;9(1):320.
19. Tu S, Zou Y, Yang M, Zhou X, Zheng X, Jiang Y, et al. Ferroptosis in hepatocellular carcinoma: Mechanisms and therapeutic implications. Biomedicine & Pharmacotherapy. 2025;182:117769.
20. Yu D, Wang Q, Zhang Q, Cai M, Liu S, Zhang W. Molecular mechanisms of ferroptosis and its antitumor applications in natural products: Ferroptosis and applications in natural products. Acta Biochimica et Biophysica Sinica. 2023;55(9):1337.
21. Xiang S, Yan W, Ren X, Feng J, Zu X. Role of ferroptosis and ferroptosis-related long non'coding RNA in breast cancer. Cellular & Molecular Biology Letters. 2024;29(1):40.
22. Wang Y, Yuan X, Ren M, Wang Z. Ferroptosis: a new research direction of Artemisinin and its derivatives in anti-cancer treatment. The American journal of Chinese medicine. 2024;52(01):161-81.
23. Sui X, Zhang R, Liu S, Duan T, Zhai L, Zhang M, et al. RSL3 drives ferroptosis through GPX4 inactivation and ROS production in colorectal cancer. Frontiers in pharmacology. 2018;9:1371.
24. Liu M, Kong X-Y, Yao Y, Wang X-A, Yang W, Wu H, et al. The critical role and molecular mechanisms of ferroptosis in antioxidant systems: a narrative review. Annals of translational medicine. 2022;10(6):368.
25. Wang D. Progress in the study of ferroptosis in cancer treatment: State-of-the-Art. Chemico-Biological Interactions. 2023;371:110348.
26. Yan H-x, Zhang Y-z, Niu Y-q, Wang Y-w, Liu L-h, Tang Y-p, et al. Investigating the interaction between calcium signaling and ferroptosis for novel cancer treatment. Phytomedicine. 2025;137:156377.
27. Stejerean-Todoran I, Gibhardt CS, Bogeski I. Calcium signals as regulators of ferroptosis in cancer. Cell calcium. 2024;124:102966.
28. Marchi S, Patergnani S, Missiroli S, Morciano G, Rimessi A, Wieckowski MR, et al. Mitochondrial and endoplasmic reticulum calcium homeostasis and cell death. Cell calcium. 2018;69:62-72.
29. Yang Y, Wang Y, Guo L, Gao W, Tang T-L, Yan M. Interaction between macrophages and ferroptosis. Cell death & disease. 2022;13(4):355.
30. Xue Q, Kang R, Klionsky DJ, Tang D, Liu J, Chen X. Copper metabolism in cell death and autophagy. Autophagy. 2023;19(8):2175-95.
31. Xie J, Su Y, Shang W, Wu Y, He J, Li T, et al. Involvement of copper in cell death and cancer. Apoptosis. 2025;30(1):488-505.
32. Baldea I, Iacoviță C, Gurgu RA, Vizitiu AS, Râzniceanu V, Mitrea DR. Magnetic Hyperthermia with Iron Oxide Nanoparticles: From Toxicity Challenges to Cancer Applications. Nanomaterials. 2025;15(19):1519.
33. Zhao Q, Qi T. The implications and prospect of cuproptosis-related genes and copper transporters in cancer progression. Frontiers in oncology. 2023;13:1117164.
34. Wang H, Yang Y, Du J. Cuproptosis: the mechanisms of copper-induced cell death and its implication in colorectal cancer. Naunyn-Schmiedeberg's Archives of Pharmacology. 2025:1-13.
35. Wang Y, Yin F, Jin Q, Liu C, Qi Z, Chen D, et al. Cuproptosis: mechanism and application in lymphoma. Current Cancer Drug Targets. 2025;25(7):766-79.
36. Lu K, Wijaya CS, Yao Q, Jin H, Feng L. Cuproplasia and cuproptosis, two sides of the coin. Cancer Communications. 2025;45(5):505-24.
37. Suwara J, Hartman ML. Balancing between cuproplasia and copper-dependent cell death: molecular basis and clinical implications of ATOX1 in cancer. Journal of Experimental & Clinical Cancer Research. 2025;44(1):222.
38. Zhang S, Huang Q, Ji T, Li Q, Hu C. Copper homeostasis and copper-induced cell death in tumor immunity: implications for therapeutic strategies in cancer immunotherapy. Biomarker Research. 2024;12(1):130.
39. Gao Q, Yin X-d, Zhang F, Zhu Y-Z, Li Z-l. The regulatory effects of traditional Chinese medicine on ferroptosis. Oxidative Medicine and Cellular Longevity. 2022;2022(1):4578381.
40. Lv H, Liu X, Zeng X, Liu Y, Zhang C, Zhang Q, et al. Comprehensive analysis of cuproptosis-related genes in immune infiltration and prognosis in melanoma. Frontiers in pharmacology. 2022;13:930041.
41. Xiaona X, Liu Q, Zhou X, Liang R, Yang S, Xu M, et al. Comprehensive analysis of cuproptosis-related genes in immune infiltration and prognosis in lung adenocarcinoma. Computers in Biology and Medicine. 2023;158:106831.
42. Patergnani S, Danese A, Bouhamida E, Aguiari G, Previati M, Pinton P, et al. Various aspects of calcium signaling in the regulation of apoptosis, autophagy, cell proliferation, and cancer. International journal of molecular sciences. 2020;21(21):8323.
43. Parys JB, Bultynck G, Vervliet T. IP3 receptor biology and endoplasmic reticulum calcium dynamics in cancer. Cellular Biology of the Endoplasmic Reticulum. 2021:215-37.
44. Danese A, Marchi S, Vitto VAM, Modesti L, Leo S, Wieckowski MR, et al. Cancer-related increases and decreases in calcium signaling at the endoplasmic reticulum-mitochondria interface (MAMs). Organelles in Disease. 2020:153-93.
45. Bonora M, Patergnani S, Ramaccini D, Morciano G, Pedriali G, Kahsay AE, et al. Physiopathology of the permeability transition pore: molecular mechanisms in human pathology. Biomolecules. 2020;10(7):998.
46. Zhai K, Liskova A, Kubatka P, Büsselberg D. Calcium entry through TRPV1: a potential target for the regulation of proliferation and apoptosis in cancerous and healthy cells. International journal of molecular sciences. 2020;21(11):4177.
47. Gu J, Guo C, Ruan J, Li K, Zhou Y, Gong X, et al. From ferroptosis to cuproptosis, and calcicoptosis, to find more novel metals-mediated distinct form of regulated cell death. Apoptosis. 2024;29(5):586-604.
48. Wang C, Peng J, Xiao Y, Zhang Z, Yang X, Liang X, et al. Advances in nanotherapeutics for tumor treatment by targeting calcium overload. Colloids and Surfaces B: Biointerfaces. 2025;245:114190.
49. Costa MI, Sarmento-Ribeiro AB, Gonçalves AC. Zinc: from biological functions to therapeutic potential. International Journal of Molecular Sciences. 2023;24(5):4822.
50. Mu S, Yang H, Wang S, Tong A, Ding R, Wang J, et al. Zinc-based Nanomaterials in Cancer Therapy: Mechanisms, Applications, and Future Directions. Theranostics. 2025;15(15):7841.
51. Tseriotis V-S, Ampazis D, Karachrysafi S, Papamitsou T, Petrakis G, Kouvelas D, et al. ZnO-Based Nanoparticles for Targeted Cancer Chemotherapy and the Role of Tumor Microenvironment: A Systematic Review. International Journal of Molecular Sciences. 2025;26(17):8417.
52. Xi Z-Y, Fan C-Y, Jiang Y-Y, Xi X-R, Nie G-Y, Zhu S, et al. Nanocatalytic system releases overloaded zinc ions and ROS to induce Znproptosis and interrupt cell cycle through inhibiting Akt/mTOR pathway. Theranostics. 2025;15(10):4734.
53. Zimta A-A, Schitcu V, Gurzau E, Stavaru C, Manda G, Szedlacsek S, et al. Biological and molecular modifications induced by cadmium and arsenic during breast and prostate cancer development. Environmental research. 2019;178:108700.
54. Nagakannan P, Tabeshmehr P, Eftekharpour E. Oxidative damage of lysosomes in regulated cell death systems: Pathophysiology and pharmacologic interventions. Free Radical Biology and Medicine. 2020;157:94-127.
55. You Y, Guo Z, Wolter T, Hu Q. Intracellular metal ion-based chemistry for programmed cell death. Chemical Society Reviews. 2025.
56. Wang J, Tian Q, Liu Y, Cai C-Y, Fu S, Li J, et al. Targeting metalloptosis in tumor therapy: from molecular mechanisms to application of metal nanoparticles. Molecular Cancer. 2025;24(1):1-61.
57. Zhang Y, Shi J, Liu X, Feng L, Gong Z, Koppula P, et al. BAP1 links metabolic regulation of ferroptosis to tumour suppression. Nature cell biology. 2018;20(10):1181-92.
58. Wang W, Green M, Choi JE, Gijón M, Kennedy PD, Johnson JK, et al. CD8+ T cells regulate tumour ferroptosis during cancer immunotherapy. Nature. 2019;569(7755):270-4.
59. Wang W, Green M, Choi JE, Gijón M, Kennedy PD, Johnson JK, et al. CD8(+) T cells regulate tumour ferroptosis during cancer immunotherapy. Nature. 2019;569(7755):270-4.
60. Wu T, Wang S, Liu Y, Bai X, Shi C. Roles and mechanisms of cuproptosis for reversing cancer therapeutic resistance. International Journal of Pharmaceutics. 2025:126267.
61. Liu J, Huang H, Zhang X, Shen Y, Jiang D, Hu S, et al. Unveiling the Cuproptosis in colitis and related carcinogenesis: a multifaceted player and theranode. Research. 2025;8.
62. Erxleben A. Mitochondria-targeting anticancer metal complexes. Current Medicinal Chemistry. 2019;26(4):694-728.
63. Dagan I, Palty R. Regulation of store-operated Ca2+ entry by SARAF. Cells. 2021;10(8):1887.
64. Wang D, Tang L, Zhang Y, Ge G, Jiang X, Mo Y, et al. Regulatory pathways and drugs associated with ferroptosis in tumors. Cell Death & Disease. 2022;13(6):544.
65. Ma X, Xiao L, Liu L, Ye L, Su P, Bi E, et al. CD36-mediated ferroptosis dampens intratumoral CD8+ T cell effector function and impairs their antitumor ability. Cell metabolism. 2021;33(5):1001-12. e5.
66. Du S, Zeng F, Deng G. Tumor neutrophils ferroptosis: a targetable immunosuppressive mechanism for cancer immunotherapy. Signal Transduction and Targeted Therapy. 2023;8(1):77.
67. Xiang D, Zhou L, Yang R, Yuan F, Xu Y, Yang Y, et al. Advances in ferroptosis-inducing agents by targeted delivery system in cancer therapy. International Journal of Nanomedicine. 2024:2091-112.
68. Gao M, Fan K, Chen Y, Zhang G, Chen J, Zhang Y. Understanding the mechanistic regulation of ferroptosis in cancer: the gene matters. Journal of Genetics and Genomics. 2022;49(10):913-26.
69. Markouli M, Skouras P, Piperi C. Impact of cuproptosis in gliomas pathogenesis with targeting options. Chemico-Biological Interactions. 2025:111394.
70. Yue Z, Wang R, Li J, Tang M, Yang L, Gu H, et al. Recent Advances in Polyoxometalate Based Nanoplatforms Mediated Reactive Oxygen Species Cancer Therapy. Chemistry–An Asian Journal. 2023;18(22):e202300749.
71. Wang Y, Wu X, Bao X, Mou X. Progress in the mechanism of the effect of Fe3O4 nanomaterials on ferroptosis in tumor cells. Molecules. 2023;28(11):4562.
72. Sant’Angelo D, Descamps G, Lecomte V, Stanicki D, Penninckx S, Dragan T, et al. Therapeutic approaches with iron oxide nanoparticles to induce ferroptosis and overcome radioresistance in cancers. Pharmaceuticals. 2025;18(3):325.
73. Masuri S, Vaňhara P, Cabiddu MG, Moráň L, Havel J, Cadoni E, et al. Copper (II) phenanthroline-based complexes as potential anticancer drugs: a walkthrough on the mechanisms of action. Molecules. 2021;27(1):49.
74. Sahoo SS, Manna D. Nanomaterial‐Triggered Ferroptosis and Cuproptosis in Cancer Therapy. Small (Weinheim an der Bergstrasse, Germany). 2025;21(12):2412462.
75. Lin Z, Monteiro‐Riviere NA, Riviere JE. Pharmacokinetics of metallic nanoparticles. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology. 2015;7(2):189-217.
76. Lin KH. Using CRISPR/Cas9 Screens to Define Organizing Principles that Govern Drug Sensitivity in Acute Myeloid Leukemia: Duke University; 2020.
77. Costa MI, Lapa BS, Jorge J, Alves R, Carreira IM, Sarmento-Ribeiro AB, et al. Zinc prevents DNA damage in normal cells but shows genotoxic and cytotoxic effects in acute myeloid leukemia cells. International Journal of Molecular Sciences. 2022;23(5):2567.
78. Wang D, Jia H, Cao H, Hou X, Wang Q, Lin J, et al. A Dual‐Channel Ca2+ Nanomodulator Induces Intracellular Ca2+ Disorders via Endogenous Ca2+ Redistribution for Tumor Radiosensitization. Advanced Materials. 2024;36(29):2401222.
79. Yan H, Talty R, Jain A, Cai Y, Zheng J, Shen X, et al. Discovery of decreased ferroptosis in male colorectal cancer patients with KRAS mutations. Redox biology. 2023;62:102699.
80. Müller F. Oncogenic KRAS renders cells resistant to ferroptosis by upregulating FSP1: Universität zu Köln; 2023.
81. Welsh SJ, Rizos H, Scolyer RA, Long GV. Resistance to combination BRAF and MEK inhibition in metastatic melanoma: where to next? European journal of cancer. 2016;62:76-85.
82. Tsymbal S, Refeld A, Kuchur O. The p53 tumor suppressor and copper metabolism: an unrevealed but important link. Molecular Biology. 2022;56(6):979-92.
83. Shi L, Liu Y, Li M, Luo Z. Emerging roles of ferroptosis in the tumor immune landscape: from danger signals to anti‐tumor immunity. The FEBS Journal. 2022;289(13):3655-65.
84. Shao C, Yan X, Pang S, Nian D, Ren L, Li H, et al. Bifunctional molecular probe targeting tumor PD-L1 enhances anti-tumor efficacy by promoting ferroptosis in lung cancer mouse model. International Immunopharmacology. 2024;130:111781.
85. Huang Q, Hu J, Chen L, Lin W, Yang J, Hu W, et al. Carbon ion radiotherapy combined with immunotherapy: synergistic anti-tumor efficacy and preliminary investigation of ferroptosis. Cancer Immunology, Immunotherapy. 2023;72(12):4077-88.
86. Bell HN, Zou W. Beyond the barrier: unraveling the mechanisms of immunotherapy resistance. Annual review of immunology. 2024;42.
87. Tian H, Duan J, Li B, Qin S, Nice EC, Zhang W, et al. Clinical chemotherapeutic agent coordinated copper‐based nanoadjuvants for efficiently sensitizing cancer chemo‐immunotherapy by cuproptosis‐mediated mitochondrial metabolic reprogramming. Advanced Functional Materials. 2023;33(51):2306584.