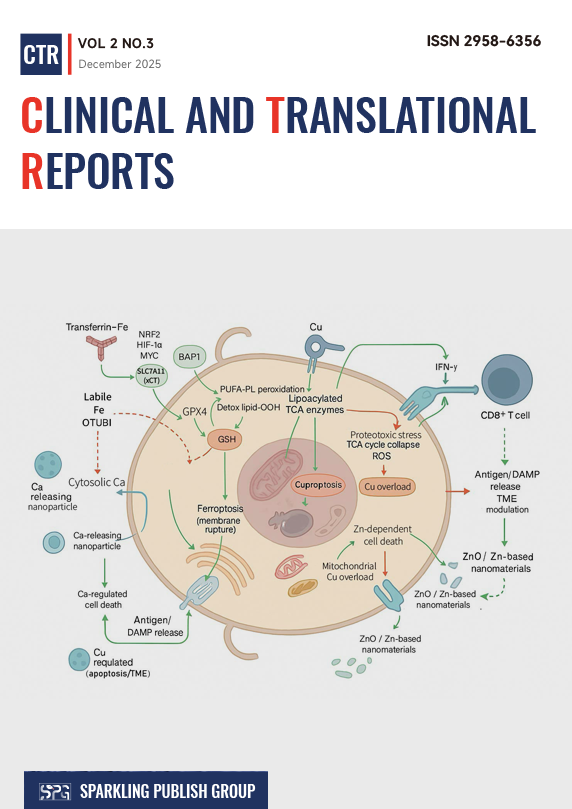
Cancer metabolism has evolved from a descriptive feature to a therapeutic target integrating oncogenic signaling, tissue constraints, and immune regulation. Metabolic reprogramming can meet biosynthetic demands under fluctuating nutrition and oxygen, maintain redox homeostasis while preserving mitochondrial function, and shape an immunosuppressive tumor microenvironment through metabolite gradients, acidity, and substrate competition. This review summarizes the major metabolic modules that sustain malignant progression, including: glucose utilization and its lactate-driven ecosystem effects; lipid metabolic remodeling that couples fatty acid oxidation to mitochondrial dynamics and translocation behavior; amino acid-dependent enhancements of thiol buffering and ferroptosis resistance; and mitochondrial energy programs that enable adaptive switching under therapeutic stress. Emerging metabolic-targeted therapies are categorized, encompassing inhibition of nutrient signaling pathways and hypoxia adaptation, lipid and amino acid-directed interventions that disrupt buffering circuits, and strategies that inhibit mitochondrial quality control. A recurring theme is that therapeutic efficacy is often limited by metabolic plasticity, which is enhanced by autophagy, stress response signaling, and synergistic adaptations among the tumor, matrix, and immune system. Therefore, delivery innovation has become a mechanistic determinant, as antibody-drug conjugates, prodrug structures, catalytic nanoreactors, and biomimetic carriers can localize metabolic stress, amplify reaction chemistry, and promote the co-localization of co-perturbations. Finally, rational combination regimens with chemotherapy, targeted therapy, and immunotherapy are discussed, aiming to prevent compensatory reprogramming while transforming the metabolically inhibited microenvironment into an immune-allowed state.
1. You M, Xie Z, Zhang N, Zhang Y, Xiao D, Liu S, et al. Signaling pathways in cancer metabolism: mechanisms and therapeutic targets. Signal transduction and targeted therapy. 2023;8(1):196. https://doi.org/10.1038/s41392-023-01442-3
2. DeBerardinis RJ, Chandel NS. Fundamentals of cancer metabolism. Science advances. 2016;2(5):e1600200. https://doi.org/10.1126/sciadv.1600200
3. Augustin RC, Delgoffe GM, Najjar YG. Characteristics of the tumor microenvironment that influence immune cell functions: hypoxia, oxidative stress, metabolic alterations. Cancers. 2020;12(12):3802. https://doi.org/10.3390/cancers12123802
4. Zou F, Zhang Y, Wang M, Quan Y, Ping H. Macrophages: Traffic centers in the tumor immune microenvironment. Cellular Immunology. 2025:105037. https://doi.org/10.1016/j.cellimm.2025.105037
5. Zhu X, Zhu L, Wu Y. Manipulating metabolic alterations and their consequences to unleash the potential of antitumor immunotherapy. Current Protein and Peptide Science. 2022;23(9):585-601. https://doi.org/10.2174/1389203723666220620161742
6. Lulla AR, Akli S, Karakas C, Caruso JA, Warma LD, Fowlkes NW, et al. Neutrophil elastase remodels mammary tumors to facilitate lung metastasis. Molecular cancer therapeutics. 2024;23(4):492-506. https://doi.org/10.1158/1535-7163.mct-23-0414
7. Zidel A, Benton A, Brown E, Shmuel S, Vanover A, Panikar SS, et al. Modulation of HER2 internalization enhances single-dose antibody-drug potency in HER2+ gastric cancer. Scientific reports. 2025;15(1):16964. https://doi.org/10.1038/s41598-025-01947-7
8. Zhu Y, Wang N, Ling J, Yang L, Omer A, Ouyang X-k, et al. In situ generation of copper (Ⅱ)/diethyldithiocarbamate complex through tannic acid/copper (Ⅱ) network coated hollow mesoporous silica for enhanced cancer chemodynamic therapy. Journal of Colloid and Interface Science. 2024;660:637-46. https://doi.org/10.1016/j.jcis.2024.01.121
9. Zhu X, Gao L, Zheng Y, Zhao PF, Wei H, Liu R, et al. Porous Fe/Cu Nanoreactor with Dual Insurance Design for Precision Chemotherapy and Chemodynamic Therapy. Advanced Healthcare Materials. 2025;14(16):2405229. https://doi.org/10.1002/adhm.202405229
10. Zou L, Liu X, Li J, Li W, Zhang L, Fu C, et al. Redox-sensitive carrier-free nanoparticles self-assembled by disulfide-linked paclitaxel-tetramethylpyrazine conjugate for combination cancer chemotherapy. Theranostics. 2021;11(9):4171. https://doi.org/10.7150/thno.42260
11. Zhu W, Yu M, Wang M, Zhang M, Hai Z. Sequential self-assembly and release of a camptothecin prodrug for tumor-targeting therapy. Nanoscale. 2025;17(4):2061-7. https://doi.org/10.1039/d4nr03519d
12. Zhu S, Sun F, Zhao P, Liang G, Sun X, Zeng L, et al. Brain-targeting biomimetic nanoparticles for codelivery of celastrol and LY2157299 for reversing glioma immunosuppression. International Journal of Pharmaceutics. 2022;619:121709. https://doi.org/10.1016/j.ijpharm.2022.121709
13. Zuberi A, Rehman U, Gupta G, Ghazwani M, Hani U, Kesharwani P. Smart Liposomal Systems for Brain Cancer: Technological Innovations in Drug Delivery. Colloids and Surfaces B: Biointerfaces. 2025:114904. https://doi.org/10.1016/j.colsurfb.2025.114904
14. Zou T, Lu W, Mezhuev Y, Lan M, Li L, Liu F, et al. A review of nanoparticle drug delivery systems responsive to endogenous breast cancer microenvironment. European Journal of Pharmaceutics and Biopharmaceutics. 2021;166:30-43. https://doi.org/10.1016/j.ejpb.2021.05.029
15. Zou T, Huang Y, Zhou Z, He S, Liu J, Chen Y, et al. A minimalist multifunctional nano-prodrug for drug resistance reverse and integration with PD-L1 mAb for enhanced immunotherapy of hepatocellular carcinoma. Journal of Nanobiotechnology. 2024;22(1):750. https://doi.org/10.1186/s12951-024-03027-w
16. Zhu X, Yu Z, Feng L, Deng L, Fang Z, Liu Z, et al. Chitosan-based nanoparticle co-delivery of docetaxel and curcumin ameliorates anti-tumor chemoimmunotherapy in lung cancer. Carbohydrate polymers. 2021;268:118237. https://doi.org/10.1016/j.carbpol.2021.118237
17. Zuo B, Wang L, Li X, Li X, Wang J, Xiong Y, et al. Abnormal low expression of SFTPC promotes the proliferation of lung adenocarcinoma by enhancing PI3K/AKT/mTOR signaling transduction. Aging (Albany NY). 2023;15(21):12451. https://doi.org/10.18632/aging.205191
18. Zohar Y, Mabjeesh NJ. Targeting HIF-1 for prostate cancer: a synthesis of preclinical evidence. Expert Opinion on Therapeutic Targets. 2023;27(8):715-31. https://doi.org/10.1080/14728222.2023.2248381
19. Zhu J-M, Sun W, Johnson TM, Lu Z, Tan D. Correction Strategies for Isotopic Ratios in Overspiked Samples Using the Double Spike Technique. Analytical Chemistry. 2024;96(50):19965-71. https://doi.org/10.1021/acs.analchem.4c04391
20. Elhady SS, Goda MS, Mehanna ET, El-Sayed NM, Hazem RM, Elfaky MA, et al. Ziziphus spina-christi L. extract attenuates bleomycin-induced lung fibrosis in mice via regulating TGF-β1/SMAD pathway: LC-MS/MS Metabolic profiling, chemical composition, and histology studies. Biomedicine & Pharmacotherapy. 2024;176:116823. https://doi.org/10.1016/j.biopha.2024.116823
21. Zhu Y, Wang Y, Li Y, Li Z, Kong W, Zhao X, et al. Carnitine palmitoyltransferase 1A promotes mitochondrial fission by enhancing MFF succinylation in ovarian cancer. Communications Biology. 2023;6(1):618. https://doi.org/10.1038/s42003-023-04993-x
22. Zuo Q, Park NH, Lee JK, Madak Erdogan Z. Liver metastatic breast cancer: epidemiology, dietary interventions, and related metabolism. Nutrients. 2022;14(12):2376. https://doi.org/10.3390/nu14122376
23. Zhu S, Zhang T, Gao H, Jin G, Yang J, He X, et al. Combination therapy of lox inhibitor and stimuli‐responsive drug for mechanochemically synergistic breast cancer treatment. Advanced healthcare materials. 2023;12(21):2300103. https://doi.org/10.1002/adhm.202300103
24. Zhu X, Li Q, Song W, Peng X, Zhao R. P2X7 receptor: a critical regulator and potential target for breast cancer. Journal of Molecular Medicine. 2021;99(3):349-58. https://doi.org/10.1007/s00109-021-02041-x
25. Zou L, Zhang T, Yang C, Liu W, Fahira A, Yang D, et al. Downregulation of SLC7A11 by Bis (4‐Hydroxy‐3, 5‐Dimethylphenyl) Sulfone Induces Ferroptosis in Hepatocellular Carcinoma Cell. Molecular Carcinogenesis. 2025;64(3):580-96. https://doi.org/10.1002/mc.23874
26. Zou Q, Zhou X, Lai J, Zhou H, Su J, Zhang Z, et al. Targeting p62 by sulforaphane promotes autolysosomal degradation of SLC7A11, inducing ferroptosis for osteosarcoma treatment. Redox Biology. 2025;79:103460. https://doi.org/10.1016/j.redox.2024.103460
27. Zou R, Hao Y, Qi C, Peng X, Huang Z, Li D, et al. Trimethyl chitosan–cysteine-based nanoparticles as an effective delivery system for portulacerebroside A in the management of hepatocellular carcinoma cells in vitro and in vivo. Journal of Drug Targeting. 2024;32(5):570-84. https://doi.org/10.1080/1061186x.2024.2344495
28. Wang J, Zhao Y-T, Sun M-J, Chen F, Guo H-L. The role of single nucleotide polymorphisms in transporter proteins and the folate metabolism pathway in delayed methotrexate excretion: a case report and literature review. Pharmacogenomics and Personalized Medicine. 2022:919-26. https://doi.org/10.2147/pgpm.s376797
29. Zhu X, Xie L, Liao Q, Tian J, Peng J, Chen Z, et al. Calcium phosphate-mineralized nanoplatform for enhanced ferroptosis and synergistic anti-PDL1 therapy in triple-negative breast cancer through multi-pathway targeting. Acta Biomaterialia. 2025. https://doi.org/10.1016/j.actbio.2025.08.014
30. Zhu Y, Chen S, Su H, Meng Y, Zang C, Ning P, et al. CPT1A-mediated MFF succinylation promotes stemness maintenance in ovarian cancer stem cells. Communications Biology. 2025;8(1):250. https://doi.org/10.1038/s42003-025-07720-w
31. Zuo L, Nie W, Yu S, Zhuang W-R, Liang C, Li S, et al. Biomimetic nanovesicle with mitochondria-synthesized sonosensitizer and mitophagy inhibition for cancer sono-immunotherapy. Nano Letters. 2023;23(7):3005-13. https://doi.org/10.1021/acs.nanolett.3c00383
32. Zhu M, Xu S, Li G, Xu G, Zhang Z, Liang H, et al. Development of a High-Efficacy and Low-Toxicity Cobalt (II) Agent for Targeting Inhibition of Tumor Growth through Mitochondrial Damage-Mediated Chemotherapy and Immunotherapy. Journal of Medicinal Chemistry. 2025. https://doi.org/10.1021/acs.jmedchem.5c01235
33. Zou P, Tao Z, Yang Z, Xiong T, Deng Z, Chen Q. The contribution of the novel CLTC-VMP1 fusion gene to autophagy regulation and energy metabolism in cisplatin-resistant osteosarcoma. American Journal of Physiology-Cell Physiology. 2024.
34. Zou L, Li X, Lin J, Fang X, Lin J, Zhang L, et al. Ethyl 2, 2-difluoro-2-(2-oxo-2H-chromen-3-yl) acetate attenuates the malignant biological behaviors of non-small cell lung cancer via suppressing EGFR/PI3K/AKT/mTOR signaling pathway. Journal of Pharmacy and Pharmacology. 2024;76(3):269-82.
35. Zughaibi TA, Suhail M, Tarique M, Tabrez S. Targeting PI3K/Akt/mTOR pathway by different flavonoids: a cancer chemopreventive approach. International Journal of Molecular Sciences. 2021;22(22):12455.
36. Zhu S, Jiao W, Xu Y, Hou L, Li H, Shao J, et al. Palmitic acid inhibits prostate cancer cell proliferation and metastasis by suppressing the PI3K/Akt pathway. Life Sciences. 2021;286:120046.
37. Zúñiga L, Cayo A, González W, Vilos C, Zúñiga R. Potassium channels as a target for cancer therapy: current perspectives. OncoTargets and therapy. 2022;15:783.
38. Yazdan M, Naghib SM, Mozafari M. Polymeric micelle-based nanogels as emerging drug delivery systems in breast cancer treatment: Promises and challenges. Current Drug Targets. 2024;25(10):649-69.
39. Zhu L-B, Xu W-L, Zhang W-W, Wu M-C, Li W-Z, Ge F, et al. De novo synthesis of pH-responsive, self-assembled, and targeted polypeptide nano-micelles for enhanced delivery of doxorubicin. Nanotechnology. 2021;32(29):295707.
40. Zu D, Yao J, Li X, Huang Y, Hu C, Deng M, et al. Dual-Targeted Biomimetic Nanoparticles for Enhanced Delivery of Polyphyllin B Synergistically Induce Ferroptosis and Immunogenic Cell Death in Gastric Cancer. ACS Applied Materials & Interfaces. 2025;17(45):61676-94.
41. Ding JF, Farah MH, Nayfeh T, Malandris K, Manolopoulos A, Hasan B, et al., editors. Targeted therapy–and chemotherapy-associated skin toxicities: systematic review and meta-analysis. Oncology nursing forum; 2020: Oncology Nursing Society.
42. Zhu P, Li T, Li Q, Gu Y, Shu Y, Hu K, et al. Mechanism and role of endoplasmic reticulum stress in osteosarcoma. Biomolecules. 2022;12(12):1882.
43. Zhu S-L, Qi M, Chen M-T, Lin J-P, Huang H-F, Deng L-J, et al. A novel DDIT3 activator dehydroevodiamine effectively inhibits tumor growth and tumor cell stemness in pancreatic cancer. Phytomedicine. 2024;128:155377.
44. Zhu Y, Tang S, Yuan Q, Fu J, He J, Liu Z, et al. Integrated characterization of hepatobiliary tumor organoids provides a potential landscape of pharmacogenomic interactions. Cell Reports Medicine. 2024;5(2).
45. Zuo W, Chen W, Liu J, Huang S, Chen L, Liu Q, et al. Macrophage-mimic hollow mesoporous Fe-based nanocatalysts for self-amplified chemodynamic therapy and metastasis inhibition via tumor microenvironment remodeling. ACS Applied Materials & Interfaces. 2022;14(4):5053-65.
46. Liu Y, Chen X, Evan T, Esapa B, Chenoweth A, Cheung A, et al., editors. Folate receptor alpha for cancer therapy: an antibody and antibody-drug conjugate target coming of age. [C]//Mabs. Taylor & Francis, 2025, 17(1): 2470309. http://doi.org/10.1080/19420862.2025.2470309
47. Tarone L, Iacoviello A, Di Lorenzo A, Verta R, Cossu C, Conti L, et al. Exploring Emerging Therapeutic Targets in Osteosarcoma by Revisiting the Immune and Cancer-Intrinsic Hallmarks of Cancer. Cancers. 2025;17(23):3846.
48. Liu X, Iovanna J, Santofimia-Castano P. Stroma-targeting strategies in pancreatic cancer: a double-edged sword. Journal of physiology and biochemistry. 2023;79(1):213-22.
49. Wang K, Zerdes I, Johansson HJ, Sarhan D, Sun Y, Kanellis DC, et al. Longitudinal molecular profiling elucidates immunometabolism dynamics in breast cancer. Nature Communications. 2024;15(1):3837.
50. Stienstra R, Netea-Maier RT, Riksen NP, Joosten LA, Netea MG. Specific and complex reprogramming of cellular metabolism in myeloid cells during innate immune responses. Cell metabolism. 2017;26(1):142-56.
51. Zhu S, Zhao Y, Xing C, Guo W, Huang Z, Zhang H, et al. Immune infiltration and drug specificity analysis of different subtypes based on functional status in angioimmunoblastic T-cell lymphoma. Heliyon. 2023;9(8).
52. Qureshi AA, Wehrle CJ, Ferreira-Gonzalez S, Jiao C, Hong H, Dadgar N, et al. Tumor organoids for primary liver cancers: A systematic review of current applications in diagnostics, disease modeling, and drug screening. JHEP Reports. 2024;6(12):101164.
53. Zhao M, Di X, Zhao Y. Plasma Proteome Profiling Identifies Biomarkers and Potential Drug Targets for Non-Small Cell Lung Cancer. International Journal of Medical Sciences. 2025;22(15):4036.
54. Jiang Z, Lu Y, Wang X. Therapeutic targets in lung diseases identified through single-cell analysis and mendelian randomization. Heart & Lung. 2025;74:211-23.
55. Huang G, Pan S-T. ROS‐Mediated therapeutic strategy in Chemo‐/Radiotherapy of head and neck cancer. Oxidative medicine and cellular longevity. 2020;2020(1):5047987.
56. Zhu S, Sun C, Cai Z, Wu J, Han X, Wang J, et al. Multifunctional nanoparticle-mediated targeting of metabolic reprogramming and DNA damage response pathways to treat drug-resistant triple-negative breast cancer. Journal of Controlled Release. 2025;381:113601.
57. Zhu S, Li J, Sun H, Liang J, Qiu Z, Zhou X, et al. A biotin guided Pt IV amphiphilic prodrug synergized with CDK4/6 inhibition for enhanced tumor targeted therapy. Nanoscale. 2025;17(16):9907-13.
58. Tong W-G, Ding X-Z, Adrian TE. The mechanisms of lipoxygenase inhibitor-induced apoptosis in human breast cancer cells. Biochemical and biophysical research communications. 2002;296(4):942-8.
59. Prendeville H, Lynch L. Diet, lipids, and antitumor immunity. Cellular & Molecular Immunology. 2022;19(3):432-44.
60. Arner EN, Rathmell JC. Metabolic programming and immune suppression in the tumor microenvironment. Cancer cell. 2023;41(3):421-33.
61. Varghese S, Pramanik S, Williams LJ, Hodges HR, Hudgens CW, Fischer GM, et al. The glutaminase inhibitor CB-839 (telaglenastat) enhances the antimelanoma activity of T-cell–mediated immunotherapies. Molecular cancer therapeutics. 2021;20(3):500-11.
62. Luby A, Alves-Guerra M-C. Targeting metabolism to control immune responses in cancer and improve checkpoint blockade immunotherapy. Cancers. 2021;13(23):5912.